M Kozminsky*, OJ Scheideler*, B Li, NK Liu, LL Sohn. “Multiplexed DNA-Directed Patterning of Antibodies for Applications in Cell Subpopulation Analysis.” ACS applied materials & interfaces 13.39 (2021): 46421-46430. *denotes equal contribution
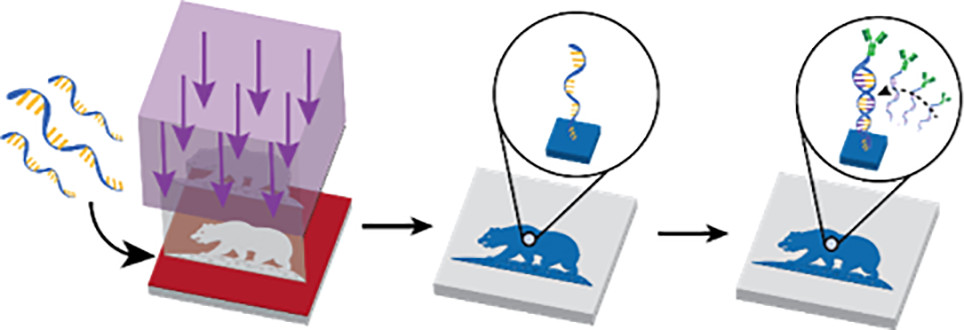
Abstract: Antibodies provide the functional biospecificity that has enabled the development of sensors, diagnostic tools, and assays in both laboratory and clinical settings. However, as multimarker screening becomes increasingly necessary due to the heterogeneity and complexity of human pathology, new methods must be developed that are capable of coordinating the precise assembly of multiple, distinct antibodies. To address this technological challenge, we engineered a bottom-up, high-throughput method in which DNA patterns, comprising unique 20-base pair oligonucleotides, are patterned onto a substrate using photolithography. These microfabricated surface patterns are programmed to hybridize with, and instruct the multiplexed assembly of, antibodies conjugated with the complementary DNA strands. We demonstrate that this simple, yet robust, approach preserves the antibody-binding functionality in two common applications: antibody-based cell capture and label-free surface marker screening. Using a simple proof-of-concept capture device, we achieved high purity separation of a breast cancer cell line, MCF-7, from a blood cell line, Jurkat, with capture purities of 77.4% and 96.6% when using antibodies specific for the respective cell types. We also show that antigen–antibody interactions slow cell trajectories in flow in the next-generation microfluidic node-pore sensing (NPS) device, enabling the differentiation of MCF-7 and Jurkat cells based on EpCAM surface-marker expression. Finally, we use a next-generation NPS device patterned with antibodies against E-cadherin, N-cadherin, and β-integrin—three markers that are associated with epithelial-mesenchymal transitions—to perform label-free surface marker screening of MCF10A, MCF-7, and Hs 578T breast epithelial cells. Our high-throughput, highly versatile technique enables rapid development of customized, antibody-based assays across a host of diverse diseases and research thrusts.